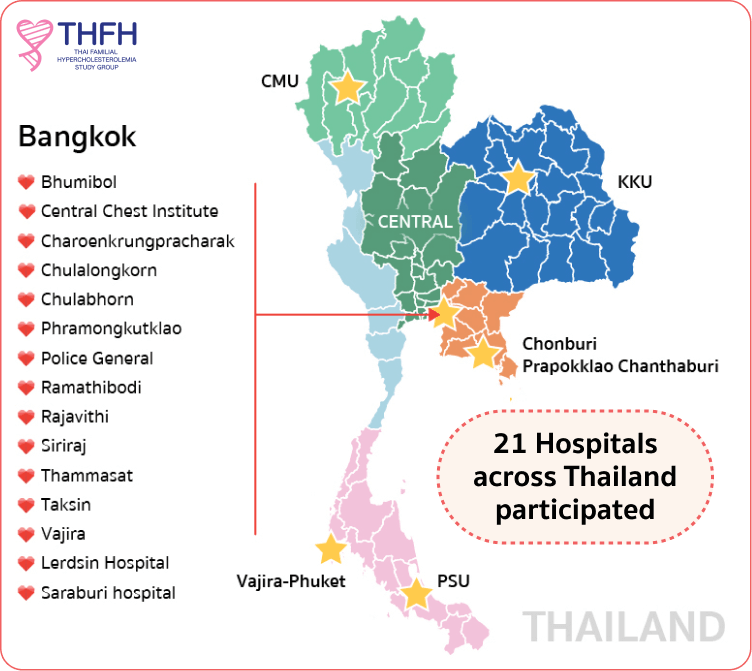
THAI FH REGISTRY
Identifying and treating patients with FH, just like what you are doing, is crucially important, but your contribution can go beyond that. Being a part of Thai FH registry is one practical way to improve care for FH patients and their families.